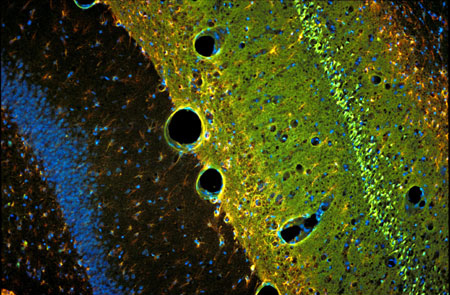
Neuroscience Research
Proteins, antibodies, kits and other reagents for neuroscience
|
Newmarket Scientific provides Antibodies, Proteins, Kits and related reagents for neuroscience research.
Some products are highlighted below, but many more are available, so please use the search box above to find specific reagents or email us at tech@nktscientific.com.
Also for more information on neuroscience research in general, including highlighted articles/papers and other resources follow our Neuroscience Twitter feed NKTSciNeuro.
|
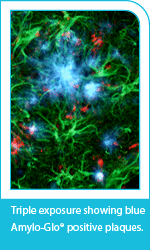
Beautifully stain amyloid plaques in tissues in multi-labelling and low magnification studies
Histologic dyes commonly used for amyloid plaque detection such as Congo Red or Thioflavin S or T come with a number of limitations, including their broad excitation frequencies which cause "bleed through" when using filters for other stains and the need for harsh chemicals during staining making them less compatible with the subsequent use of antibodies.
Amylo-Glo RTD (Cat. No. TR-300-AG) is a histological marker that shows clear superiority for visualising amyloid plaques over conventional markers with its... read more
|
||
|
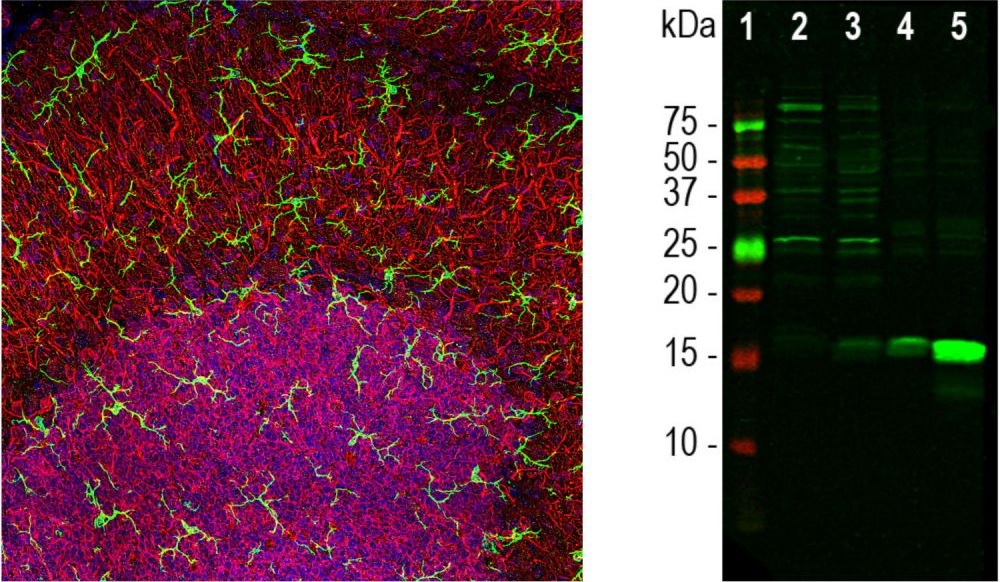
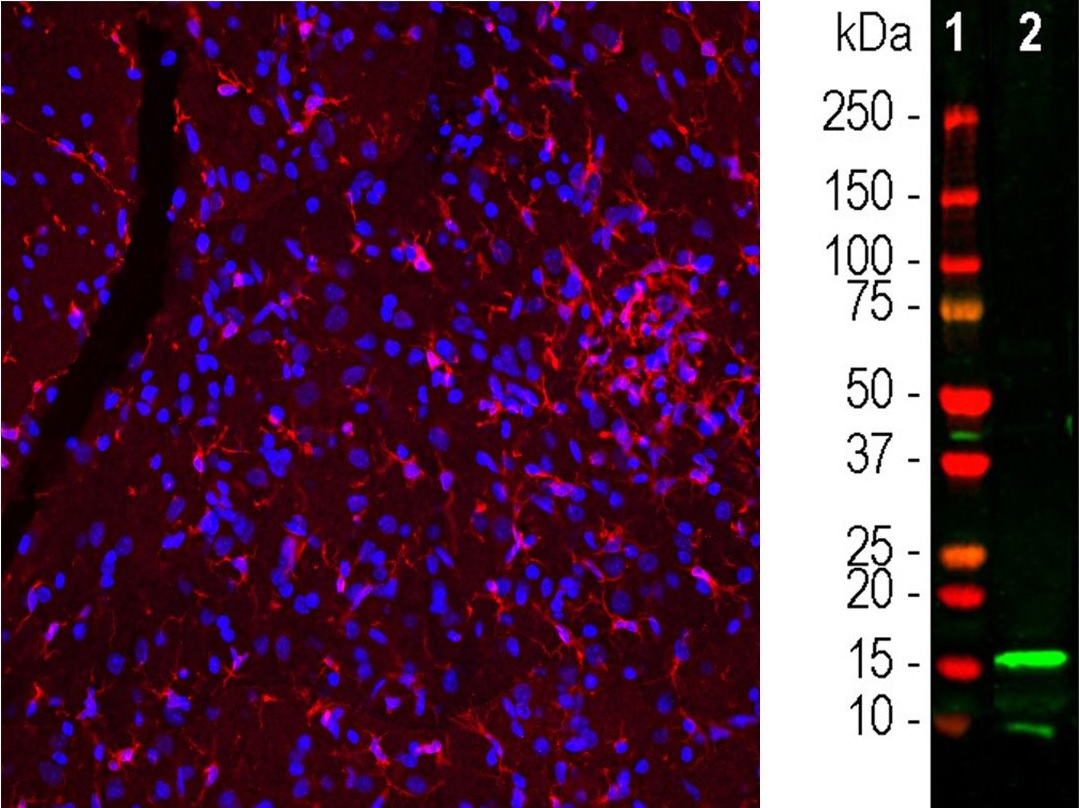
Microglia are specialised macrophages found in the central nervous system (CNS).They play a key role in brain maintenance and clear cellular debris, plaques, dead neurons and synapses, and infectious agents.
Commonly used markers are IBA1, CD11b, CD45 as well as P2RY12; a new selective marker of microglia.
|
Newmarket Scientific is the UK and Ireland Distributor of the ImmunoStar antibodies for neuroscience research. This set of antibodies was originally provided by DiaSorin (INCSTAR), before becoming ImmunoStar in 2001.
Having been available for many years this set of highly cited antibodies really has been tried and tested! Additionally ImmunoStar antibodies are put through extensive testing before release to ensure excellent reliability and lot-to-lot consistency.
Get further details of the full range of ImmunoStar antibodies for neuroscience research.
|
||
|
Fluoro-Jade C stains all degenerating neurons regardless of the cause of the damage or the mechanism of cell death. Due to its low background and high resolution it provides high contrast staining, making it ideal for localising degenerating nerve cell bodies as well as distal dendrites, axons and terminals. Fluoro-Jade C is resistant to fading making it compatible with most histological processing and staining protocols.
Fluoro-Jade C (FJC) Ready-to-Dilute Staining Kit - 40 mL Fluoro-Jade C (FJC) Ready-to-Dilute Staining Kit (Trial size) - 20 mL (This product is equivalent to discontinued product AG325 from Merck-Millipore)
(This product is equivalent to discontinued product AG310 (FJB) from Merck-Millipore)
|